The average adult makes upwards of 35,000 decisions a day. These can include the bigger, more existential questions that require reflection to weigh the pros and cons. But the vast majority of decisions seem insignificant: What will I wear today? Order takeout or make food at home? Podcast or playlist? Still, these fleeting impulse choices can play just as big a role in our lives as the more thought-out ones.
Prime examples of this were recently made on Reddit, when people were asked to share a "decision you made in under 10 seconds that changed your life forever." These seemingly insignificant choices changed fates in profound ways.
“Decided to go to Subway instead of Dairy Queen. They were across the street from each other and I was passing through town during lunchtime. Ended up hitting it off with the woman making my sandwich. Next week is our 11 year anniversary.”
“My best friend said she wanted to move 2000+ miles across the country back to her home state and asked me if I wanted to come. I didn’t think, said ‘Yup, I’ll go.” We moved, less than a year afterwards I met my now wife. Couldn’t be happier.”
“Said yes to adopting a stray dog that followed me home. 10-second 'sure why not' moment—now he's my best buddy for 8 years.”

A van travels down the road.
Photo credit: Canva
Others were able to uncover new passions they never imagined.
“I flipped a coin to decide if I was going to quit my job. Heads. I quit. A friend saw my lights on that night and stopped by to see what I was doing. I told him what happened, and he told me it was great timing. They let someone go at his job that day. He set me up with an interview for the next day, and I was hired. There were only three people who worked there. I eventually became the plant manager and have been working in management ever since!”
“Saw a random advert advertising scuba diving certification. I signed up thinking ‘why not?’ I’m now an aspiring diving instructor!”
“Early 20s and my sister asked me to drive her to the music shop to buy a guitar. I point one out and say ‘that black and gold one is gorgeous. Get that one.’ She tries it out and says ‘ehhh, I dunno if I really feel this one.’ I tell her that if she doesn't buy it, I will, and she says ‘you don't even play guitar! What are you gonna do with it?’ Walked out of the store with it on a complete whim, spent 3 months learning before I started doing open mics, making friends at the music shop, joining a band, and having the time of my life throughout my 20s.”

A person plays the guitar.
Photo credit: CanvaFor some, a 10-second decision ended up with an unexpected windfall.
“Decided not to get into an elevator with my ex and her new boyfriend, so I took the stairs instead. On the third flight, I found a discarded scratch-off ticket that ended up being worth $50,000. It’s the only time in my life where being socially awkward actually paid off my mortgage.”
“A family in my marina announced that they are moving away the following week. They were going to turn their sailboat over to a broker to sell it for them. I mentioned that I was contemplating a larger boat with a smaller engine and would be interested in theirs. I asked what price would they consider? He said, $10k. I said, okay, and we shook on it. The boat was worth over $24k. I got a wheelbarrow from the marina corral and removed stuff from my smaller boat, walked it over to their dock, and loaded it onboard. That was ten years ago, and I still live on it six months out of the year. Sweet.”
Sometimes, these kinds of life-altering changes are simply fresh new outlooks on life.
“I was going through old text messages with my then girlfriend and realized I had become a very negative person. I decided right there to always look for the bright side of things. It takes some effort but that was 12 years ago and I'm much happier. It becomes second nature after a while.”

A man smiles.
Photo credit: CanvaIn many instances, a 10-second decision prevented tragedy for themselves or others.
“Most of these answers are super happy and mine really isn’t but fits the question. Mine would be double checking on my wife before bed. She suffers from PTSD, depression and anxiety and had just been ‘off’ all day. I checked with her a couple times and she said everything was alright each time. It might sound weird but she was too happy and calm but kind of sad at the same time. It just didn’t sit right with me. She said she was going to bed and we kissed and I asked again she chucked and said to stop worrying. She went upstairs and I waited a few seconds and went up to say I think something is wrong. I stopped her from committing suicide that night. Normally I would take her for her word but that decision was 12 years ago and we are having the best version of our lives because I listened to my gut and went and checked.”
“One night I drove home from working evening shift (like 2 miles) and when I glanced in the rear view I had chills when I saw the headlights behind me at a red light. I got to my house and was about to park but then heard a voice in my head tell me to keep driving, so I did… and they continued to follow me. Called my dad to stay on the phone as I drove to a police station. Car stopped following when a cop car pulled up behind us (coincidentally). Next morning read about an armed carjacking in the area about an hour later.”
“In middle school my best friend invited me to Knott's Berry Farm with another group of kids he was friends with…I got to my friend’s house after running some errands with my mom. There were 5 kids aged 14-15 there. I asked my friend how we were getting [to Knott's Berry] and he said he was driving his dad’s car (he was underage and had no license). I was nervous but I wanted to look cool so, I said ok. Right before we got into the car, I had the worst feeling come over me. In approximately 5 seconds, I felt a drop in my stomach to a bottomless pit, a hot flash followed by chills, and intense nausea. I literally left without making a sound, running to a nearby Starbucks and using a stranger's phone and asking my mother to pick me up. I felt so embarrassed. I knew I was going to get shit all month from my friend…A few hours later, they were on their way home on the freeway and lost control of the vehicle. All 5 died.”

A damaged vehicle sits on the side of a road.
Photo credit: CanvaWhat about when you can't decide?
And yet, for some (make that many) of us, knowing that every decision is uniquely important can be a major source of anxiety, causing us to freeze via “analysis paralysis” and not choose at all—which, in itself, is a choice (they’re inescapable!). In fact, our brains tend to struggle more with less risky decisions.
Interestingly, experts seem to suggest combating this ambivalence with tools that force a quick deadline. You can either toss a coin or set a time limit, which would arguably incite one of these potentially life-changing 10-second decisions.
Life will inevitably call on us to make both long, thought-out decisions and to go with our gut. But hopefully, this reminds us that even our whims can lead to something truly pivotal without making us lose our minds.



 Batteries being recycled at WRWA, London. Nov ‘21Photo by John Cameron on Unsplash
Batteries being recycled at WRWA, London. Nov ‘21Photo by John Cameron on Unsplash


 A Baby Boomer garage filled with stuff.
A Baby Boomer garage filled with stuff.

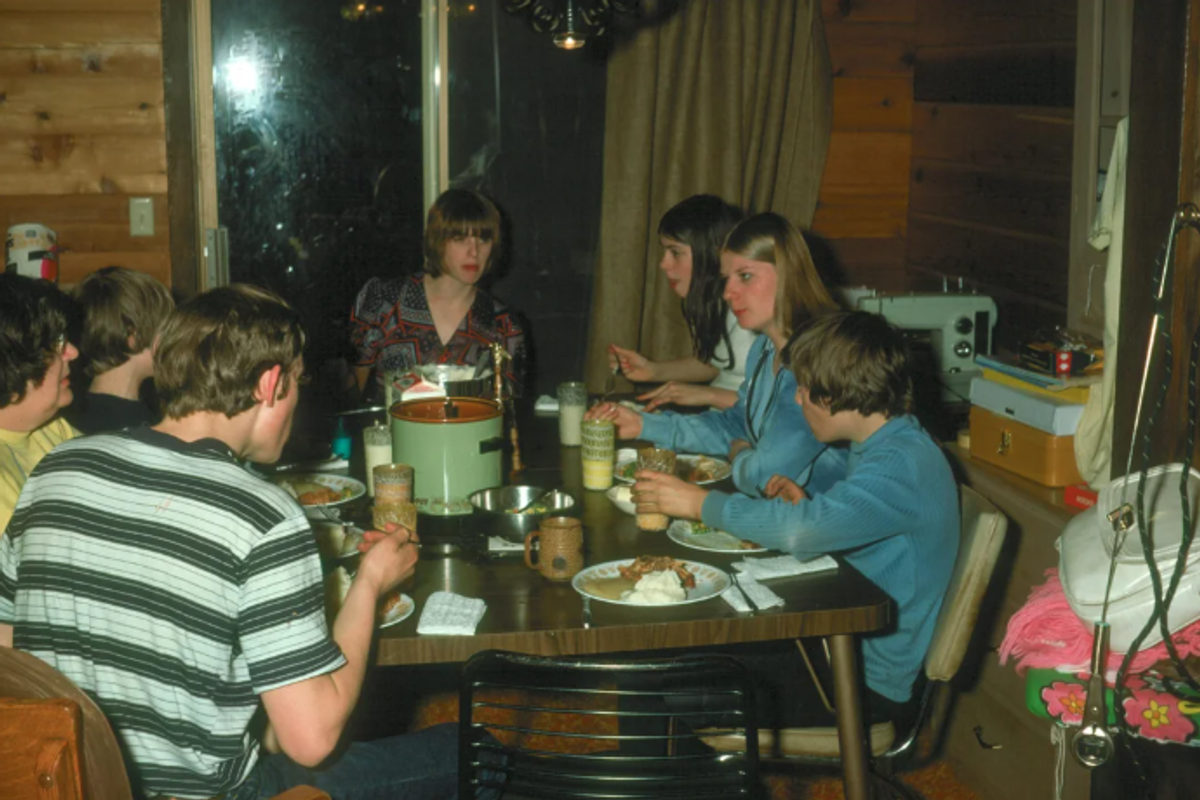

 A van travels down the road.
A van travels down the road.  A person plays the guitar.
A person plays the guitar.  A man smiles.
A man smiles.  A damaged vehicle sits on the side of a road.
A damaged vehicle sits on the side of a road. 